Difference between revisions of "Optical Loading of Magnetic Traps"
| Line 15: | Line 15: | ||
Currently, we are pursuing co-trapping of calcium monohydride (CaH) molecules with lithium (Li) atoms for studying Li-CaH collisions. We chose to study CaH because of its good collisional properties. The elastic to spin-depolarization collision ratio | Currently, we are pursuing co-trapping of calcium monohydride (CaH) molecules with lithium (Li) atoms for studying Li-CaH collisions. We chose to study CaH because of its good collisional properties. The elastic to spin-depolarization collision ratio | ||
| − | between CaH and He | + | between CaH and He was measured to be larger than 10^5. Theoretical study shows the main |
spin-depolarization mechanism of doublet sigma molecules during collisions with Helium is due to mixing | spin-depolarization mechanism of doublet sigma molecules during collisions with Helium is due to mixing | ||
of the molecular wavefunction between rotational ground and excited states. The spin-rotational coupling in the rotational excited state can cause spin-depolarization. Since CaH | of the molecular wavefunction between rotational ground and excited states. The spin-rotational coupling in the rotational excited state can cause spin-depolarization. Since CaH | ||
Revision as of 13:10, 9 February 2014
Contents
People
Post Docs
- Hsin-I Lu
- Boerge Hemmerling
Grad Students
- Ivan Kozyryev
- Louis Baum
Undergrad Students
- Michael Casson
Overview
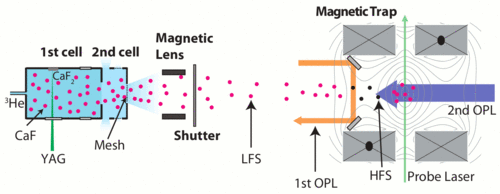
General approaches for delivering cold, chemically diverse molecules in large quantities could have a profound impact on research in quantum simulation, cold controlled chemistry, and particle physics. Buffer gas loading of polar molecules into magnetic traps has been demonstrated with many species including CaH and NH. However, increasing phase space density via evaporative or sympathetic cooling was previously inhibited by collisions with residual helium (He) buffer gas. In the current experiment, we produce a cold and slow calcium monofluoride (CaF) molecular beam with initial forward velocity around 30 m/s and load CaF (v=0, N=1) into a deep superconducting magnetic trap combining magnetic deceleration and optical pumping. A magnetic lens is used to collimate low-field seeking states. Irreversible trap loading is achieved using two optical pumping stages, where scattered photons remove molecular potential energy and entropy. Since the trap loading scheme requires scattering of only a few photons, the method is applicable to a wide range of magnetic species, including polyatomic molecules.
Currently, we are pursuing co-trapping of calcium monohydride (CaH) molecules with lithium (Li) atoms for studying Li-CaH collisions. We chose to study CaH because of its good collisional properties. The elastic to spin-depolarization collision ratio between CaH and He was measured to be larger than 10^5. Theoretical study shows the main spin-depolarization mechanism of doublet sigma molecules during collisions with Helium is due to mixing of the molecular wavefunction between rotational ground and excited states. The spin-rotational coupling in the rotational excited state can cause spin-depolarization. Since CaH has large rotational splitting (12K between N=0 and N=1 states), we expect it to maintain its spin orientation during collisions with other S state atoms. In addition, magnetic dipolar relaxation should be comparable to collisions between alkali atoms due to its moderate magnetic dipole moment. These properties make it a good candidate for sympathetic cooling of molecules.
Recent Publications
- Magnetic Trapping of Molecules via Optical Loading and Magnetic Slowing. H.-I Lu, I. Kozyryev, B. Hemmerling, J. Piskorski, and J. M. Doyle. arXiv:1310.2669 [physics.atom-ph] (2013).
- Cold and Slow Molecular Beam. Hsin-I Lu, Julia Rasmussen, Matthew J. Wright, Dave Patterson, and John M. Doyle. Phys. Chem. Chem. Phys., 2011, DOI: 10.1039/c1cp21206k.
- Intense atomic and molecular beams via neon buffer-gas cooling, D. Patterson, J. Rasmussen, and J.M. Doyle. New Journal of Physics 11, 055018 (2009).
- Bright, Guided Molecular Beam with Hydrodynamic Enhancement, D. Patterson and J.M. Doyle. J of Chem Phys 126, 154307 (2007).